- Winter 2016
- Volume 15
- Issue 1
Brain Trust: Cutting-Edge Treatments for Glioblastoma on the Rise
New treatments, such as immunotherapy, are continuing to prove successful for patients with glioblastoma, one of the most dangerous cancers.
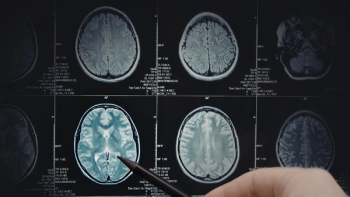
Ryan DeGrand has had no sign of glioblastoma since he started taking the experimental targeted vaccine Rintega (rindopepimut) more than 10 years ago. PHOTO BY ERIN STUBBLEFIELD
Ryan DeGrand was 33 years old and married with two young children when he started suffering debilitating headaches in 2004. The diagnosis, stage 4 glioblastoma, didn’t offer much hope — the standard regimen of surgery, chemotherapy and radiation might buy him a year, his doctors in St. Louis told him. So after he completed the treatment, he got on the Internet and began looking for research centers that specialized in treating his type of brain tumor.
That search brought him to Duke University, where researchers were in the early stages of testing a vaccine that targets EGFRvIII — a genetic abnormality of the epidermal growth factor receptor (EGFR) that occurs in about one-third of glioblastoma patients and that’s associated with tumor growth and resistance to therapy. The condition is not inherited, but arises from an acquired mutation. DeGrand’s tumor tested positive for the mutation, and he was entered into the trial.
Today, DeGrand is cancer-free and still on the vaccine, which is a series of four shots in the upper thigh that he receives once a month. “For my family, I was willing to try anything,” says DeGrand, who has started a sporting goods business and welcomed a third child since entering the trial. “I just got my MRI, and everything is clean. I’m living every day to its fullest.
About 15 percent of brain cancers are GBMs, aggressive tumors that rarely spread to other organs but that do grow rapidly inside the brain. They typically cause headaches and nausea, but if they’re located near the brain’s most critical structures, they can also trigger memory loss, weakness and speech problems. Their prevalence increases with age; only about 3 percent of childhood brain cancers are GBMs. Scientists have not yet figured out what causes this type of brain tumor.
The standard therapy for GBM starts with surgery to remove as much of the tumor as possible, as well as to pinpoint the stage of the disease, and to identify genetic markers that might point to personalized treatments. Patients then typically receive radiation and the chemotherapy drug temozolomide. Although this regimen can be very effective at shrinking the tumors, it can cause side effects like nausea, hair loss and headache. The GBMs tend to recur within a median time of seven months, and median survival time is about 15 months, although there is considerable variation.
Tackling Tumor Variations
GBMs contain a wide mix of cell types that often differ from patient to patient and even within an individual patient’s tumor, and that vary widely in their response to treatment. What’s more, when they recur after treatment, “the molecular profile of the cancer changes dramatically,” says Mark Gilbert, a senior investigator and chief of the Neuro-Oncology Branch at the National Cancer Institute. “And the further away the recurrence is from the primary tumor, the less alike it is to its parent.” Such variability in GBM tumors “makes treating them incredibly complicated,” he adds.
That’s why drugs that enable the immune system to recognize GBM tumors as enemies — and then destroy them — are considered especially promising. One of the leading contenders is the vaccine that saved DeGrand’s life, Rintega (rindopepimut), which is completing phase 3 trials (Editor's note on March 10, 2016: Despite excitement, the drug
The EGFRvIII mutation that Rintega targets is particularly attractive because it occurs only in cancer cells, says one of its discoverers, John Sampson, chief of the department of neurosurgery at Duke. And in GBM, the mutation is consistent across patients. “Some mutations can be different from patient to patient, even if they’re in the same gene, but this is the exact same mutation each time,” he says. That makes it easier to target with drugs.
Other drugs that stimulate the immune system to fight cancer are also in clinical trials in GBM, including “checkpoint inhibitors” that target immune-suppressing proteins such as PD-1 and CTLA-4. Opdivo (nivolumab) and Yervoy (ipilimumab) — which are already on the market and have been particularly effective in treating melanoma — are now being tested together in a phase 3 trial in patients with recurrent GBM. In a small phase 2 trial, the overall survival rate among patients taking the combination treatment was 80 percent after six months, versus 70 percent in patients taking Opdivo alone.
Brain cancer experts agree that, ultimately, immunotherapies may work better in combination with other immune-boosting drugs or with medicines that work differently in the brain. In 2009, for example, the FDA approved Avastin (bevacizumab) for use in patients with recurrent GBM. The drug is anti-angiogenic, meaning it chokes off the blood vessels that feed tumors. Even though it hasn’t been shown to greatly enhance overall survival rates on its own, Avastin is showing some promise in combination with newer contenders like Rintega. In a phase 2 trial in patients with recurrent GBM, 25 percent of patients who were put on Avastin plus Rintega were still alive after two years, versus none of the patients who took Avastin alone.
Avastin helps control brain edema, or swelling, which may enhance the ability of immune-boosting drugs to do their jobs. “Inducing an immune response in the brain can cause swelling,” which can, in turn, touch off a range of problems, from pain to speech difficulties, says Santosh Kesari, professor and chair of the Department of Translational Neuro-Oncology and Neuro- Therapeutics at the John Wayne Cancer Institute at Providence St. John’s Health Center in Santa Monica, California. “Combining immunotherapies with Avastin seems to be well tolerated and may be synergistic, since Avastin has an anti-edema effect.”
Bolstering the Immune System
Another potential way to tackle GBMs is with chimeric antigen receptor T-cells (CAR T cells), immune-boosting cells that are taken from individual patients, genetically engineered and reinfused. The genetic engineering equips the T cells to recognize specific tumor-associated proteins on tumor cells; this draws the T cells to the tumor cells, which they eliminate. The approach, which is in clinical trials, is furthest along in blood cancers like leukemia and lymphoma, but there’s an increasing interest in using CAR T cells to target solid tumors.
In GBM, researchers led by a team at the University of Pennsylvania have figured out how to engineer T cells to target EGFRvIII, in the hopes of shrinking tumors that have that gene mutation. An early trial of the CAR T cells is underway in patients with recurrent GBM who test positive for EGFRvIII.
David Reardon, clinical director of the Center for Neuro-Oncology at the Dana-Farber Cancer Institute in Boston, believes the CAR T cell approach is exciting, but particularly challenging in GBM. “One limitation is it hits a single mutation, and often tumors can adapt,” Reardon says. “When patients relapse, the tumor cells often don’t express EGFRvIII.” Researchers at Dana-Farber hope to clear that hurdle by making personalized vaccines for patients based on the specific mutations their GBM tumors possess. The technique involves sequencing the genome of each patient’s tumor, then using algorithms to predict which mutated genes are likely to produce proteins that would trigger a strong immune response, Reardon explains. Each vaccine contains 20 peptides, or proteins, that match the ones pumped out by those mutated genes, increasing the amount circulating through the patient’s body and hopefully attracting the attention of the immune system to both the infused proteins and to the tumor. “We give those peptides back to the patient to sensitize the immune system,” says Reardon, who reports that Dana-Farber is currently testing the strategy in a small group of patients. “Any tumor cell that has any of those 20 peptides should be attacked and eradicated. It’s a strategy for exploiting the heterogeneity in these cancers.”
A similar approach is being investigated by ImmunoCellular Therapeutics, which has developed a therapy that seems to work best in the 40 to 50 percent of GBM patients whose immune systems make HLA-A2 — a substance that generates an immune response, says Patrick Wen, the principal investigator for the phase 2 trial and director of the Center for Neuro-Oncology at Dana-Farber Cancer Institute. The treatment, called ICT- 107, uses a patient’s own immune cells, called “dendritic” cells, pulsed with six manufactured antigens known to be expressed in GBM, four of them on GBM stem cells. The aim is to teach each patient’s immune system to recognize and fight the antigens.
A randomized phase 2 study whose results were presented in November compared patients taking temozolomide plus ICT- 107 with patients taking chemotherapy plus placebo. Of those patients who were HLA-A2-positive, 60 percent treated with ICT-107 demonstrated a statistically significant immune response compared with only 36 percent of control patients. Patients whose tumors were naturally resistant to temozolomide experienced a median overall survival of 15.8 months on ICT-107 plus temozolomide, compared with 11.8 months on the chemotherapy alone. Those with tumors responsive to temozolomide experienced a median overall survival of 37.7 months on the combination versus 23.9 months on temozolomide alone.
The company is now launching a large international phase 3 study of ICT-107 in HLA-A2-positive patients at multiple sites.
Viruses that are weakened so that they don’t cause illness may also hold promise in treating GBM. Some viruses naturally infect only cancer cells, and others can be engineered to do so; scientists can also equip such a virus with a cancer-killing gene or medicine. Viruses are intriguing to scientists because they assist the immune system in fighting cancer from two different angles: The viruses actually infect cancer cells and destroy them directly, and then they stimulate the immune system to continue attacking tumors even after the viruses are cleared from the body.
Several types of engineered viruses, including herpes simplex virus and adenovirus, are in early-stage testing for GBM. And scientists at Duke are testing a form of the polio virus in patients with the disease.
Some patients have achieved long-term remissions using the polio virus, but Duke’s Sampson says quite a bit more research will be required to determine exactly how the virus is working against the cancer and how it can be optimized to enhance the benefits and lessen the risk that the treatment itself will touch off dangerous immune responses.
After chemotherapy, radiation and surgery, Jeffrey Donahue’S glioblastoma tumor can no longer be detected on MRI scans. PHOTO BY ROB ANDREW
A Better Standard of CareImproving upon existing chemotherapy and radiation methods, in particular with less toxicity, is also a priority in GBM, and there have been some encouraging advances there. In 2011, the FDA approved Optune, a device from Novocure that’s worn on the head 18 hours a day and that delivers low-intensity electrical fields directly to tumors. The therapy, a new technology never used before in cancer, specifically disrupts cancer cells’ ability to divide and multiply. Optune was initially approved for patients with recurrent GBMs along with chemotherapy, in the case of a recurrence following surgery, radiation and chemotherapy, but in October 2015, the approval was expanded to include newly diagnosed patients who have had surgery, radiation and possibly chemotherapy, but no recurrence.
Optune, which is typically given along with temozolomide, reduced the risk of death by 31 percent compared with chemotherapy alone in updated results of a clinical trial presented in late November 2015. Two-year survival without the device was just 29 percent versus 43 percent in the patients who used it. “Progression-free survival also improved significantly with the device,” says Kesari, who was one of the investigators for the clinical trial.
In addition to incorporating Optune into the standard of care, oncologists are investigating methods for predicting which patients are most likely to respond to temozolomide. In GBM, an enzyme, MGMT, can be present that makes tumors resistant to temozolomide. But about 45 percent of GBM tumors have a promoter gene that suppresses MGMT (this is called a methylated MGMT gene), making temozolomide more likely to work against the cancer. In a recent study, patients with that gene had two-year survival rates of 46 percent, versus just 14 percent among patients without it. Temozolomide is still the standard of care, Kesari says, but “we’re at the point where we’re thinking we shouldn’t give it to patients who really won’t respond to it. Instead, we should try something else along with radiation.”
Other types of chemotherapy are being studied for treating GBM, including Tasigna (nilotinib), a Novartis drug that is currently approved to treat leukemia. Tasigna targets platelet derived growth factor receptor (PDGFR), a protein that’s also prevalent in some GBM tumors.
Removing as much of the tumor prior to chemotherapy is essential for improving outcomes, and there have been several advances there, as well. Surgeons are now able to use fluorescent markers that cause tumor cells to glow, so they can better distinguish between cancerous cells and normal tissues. And image-guidance systems are integrating the growing understanding of the brain’s anatomy, so surgeons can determine how best to remove as much of a tumor as possible without affecting movement, memory, speech and other vital functions.
“Image guidance essentially allows us to have a GPS unit for the brain,” says Michael Lim, director of brain tumor immunotherapy and associate professor of neurosurgery at Johns Hopkins. “It improves the amount of tumor we can remove while ensuring we’re not causing further injury.” Studies suggest that if 80 percent or more of the tumor can be removed, survival significantly improves, Lim adds.
Such improvements in the standard of care have been a boon to patients like Jeffrey Donohue, who was 67 when he was diagnosed with GBM in 2014. The chemotherapy, radiation and surgery shrank his tumor to the point where it can no longer be detected on MRI scans. The California resident says he was left with a bit of aphasia, which is a tendency for his speech to lag behind his thoughts. But he’s grateful that advances in GBM treatments — along with his dedication to a healthy diet, exercise and meditation — have given him a second chance at life.
“The statistics were grim, but rigorous treatment put me in recovery mode,” Donahue says. “I told my doctors I plan to live to age 96.”
Articles in this issue
almost 10 years ago
Heal: The Vital Role of Dogs in the Search for Cancer Curesalmost 10 years ago
Men and Women Use Different Strategies to Choose Cancer Treatmentsalmost 10 years ago
What Causes Cancer? Three Main Culpritsalmost 10 years ago
Facing a Fierce Opponent, and Gaining Groundalmost 10 years ago
A Cancer Survivor's Menualmost 10 years ago
HPV Vaccine Uptake Low Among Boysalmost 10 years ago
Comments From Readers on CURE's Winter Issuealmost 10 years ago
Medical Illustration: Novel Treatments for Glioblastomaalmost 10 years ago
On the Road to Recovery: Traveling for Cancer Treatmentalmost 10 years ago
Getting Warmer: Women Can Revive Sexual Function After Cancer