Articles by Brielle Urciuoli

PARP inhibitors have drastically changed the treatment landscape – and outcomes – of patients with ovarian cancer. However, despite promise shown among this class of drugs, it is still unclear which agent is the best to use in the maintenance setting.

It’s been well-noted that excessive alcohol consumption can lead to a slew of health effects, including an increased risk of multiple types of cancers, but what about the effects of moderate drinking?
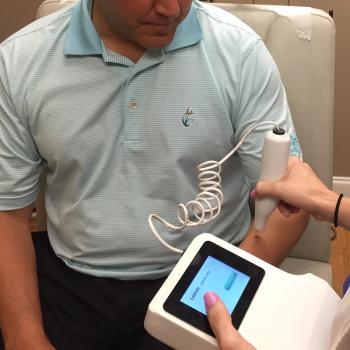
Patients and providers can evaluate lesions within seconds by running the tip of the device over their skin, and then link the results to a user-friendly smartphone app.

Among 926 patients surveyed, where the median age was 58, 66 percent had previously used marijuana. Further, 24 percent used it in the last year and 21 percent in the past month.

For people with multiple myeloma, dollars spent on treatment are being stretched farther than they used to be, a recent study shows.

Mail-order kits are now making genetic testing more accessible to patients who may need it the most.

From fatigue to chemo brain, Curetoday.com and CURE Magazine offer a variety of resources for people dealing with these effects.
In early 2015, Zika outbreaks caused widespread panic in many parts of North and South America. However, now researchers are exploring the possibility of putting the virus to good use: fighting glioblastoma multiforme (GBM), the most deadly type of brain cancer.
Women with ovarian or breast cancer often don’t receive the genetic testing they need in order to devise the best treatment plans, a new study says.

A higher percentage of body fat – specifically fat in the abdominal area – was associated with an increased risk of certain cancers for postmenopausal women, according to a recent study presented at the 2017 European Society of Medical Oncology (ESMO) Congress in Madrid, Spain.
Women with breast cancer who are positive for the BRCA gene mutation may be more likely to encounter difficulty with fertility preservation compared to women with breast cancer who have tested negative for the BRCA mutation.
Patients with advanced gastric cancer tend to use their health care resources more often and have a worse quality of life than those without the disease

While many people associate the strategy with the end of life, studies have shown that employing palliative care earlier in the course of treatment can benefit patients’ outcomes and quality of life.

Side effects that arise months or years after adolescents and young adults (AYAs) have completed cancer treatment — known as late effects — can significantly affect both careers and quality of life in this population.
Nausea and vomiting are no longer top concerns for patients undergoing chemotherapy. Socio-psychological concerns, instead, have jumped to the forefront.

A recent study looked at the financial effects that many survivors of childhood cancer face.

Although they bring devastation to the whole community hit, catastrophes, such as Tropical Storm Harvey and Hurricane Irma, introduce a new level of difficulty for patients who are being treated for cancer.

Oncology care centers in Tropical Storm Harvey’s path close down locations, provide off-site care and update patients via their websites and social media.

CURE spoke with Rami S. Komrokji, M.D. about collaborative efforts that will lead to new advances in the treatment of myelodysplastic syndrome (MDS).

Another case has been settled regarding Johnson & Johnson's Baby Powder and its possible link to ovarian cancer.

A recent study found that gender makes a difference in the survival of a patient with ET.

Researchers at Yale compared people who used conventional therapy to those who used only alternative methods
Driven by personal experience and his love for art, Ryan Berardi created a 3D animation that would help health care professionals if they wrongly administer chemotherapy to patients.
Can a drug used to treat arthritis also be a way to treat polycythemia vera (PV), and save patients with the blood cancer money?

A recent study found that children of older parents may have an increased risk of certain types of cancers.

For years, people believed that there was a link between vasectomies and prostate cancer.

A recent study found that women with breast cancer often have a network of people that they consult to help them make treatment decisions.

"I wanted to go do it the best I could, considering all the physical limitations. I just wanted to start, and I wanted to finish the Boston Marathon. The notion of time was not in the equation."
— JEFF GOAD, multiple myeloma survivor

Chemotherapy-induced peripheral neuropathy affects many patients with cancer, so CURE spoke with an expert about coping strategies for those who have developed this condition.

Ovarian cancer screening for women who do not have symptoms of the disease may do more harm than good, according to the USPSTF.



































