Article
Encouraging Trial Results Announced for Breyanzi in Lymphoma Treatment
Author(s):
Breyanzi has reportedly shown promising effects for patients with relapsed or refractory follicular lymphoma and B-cell non-Hodgkin lymphoma.
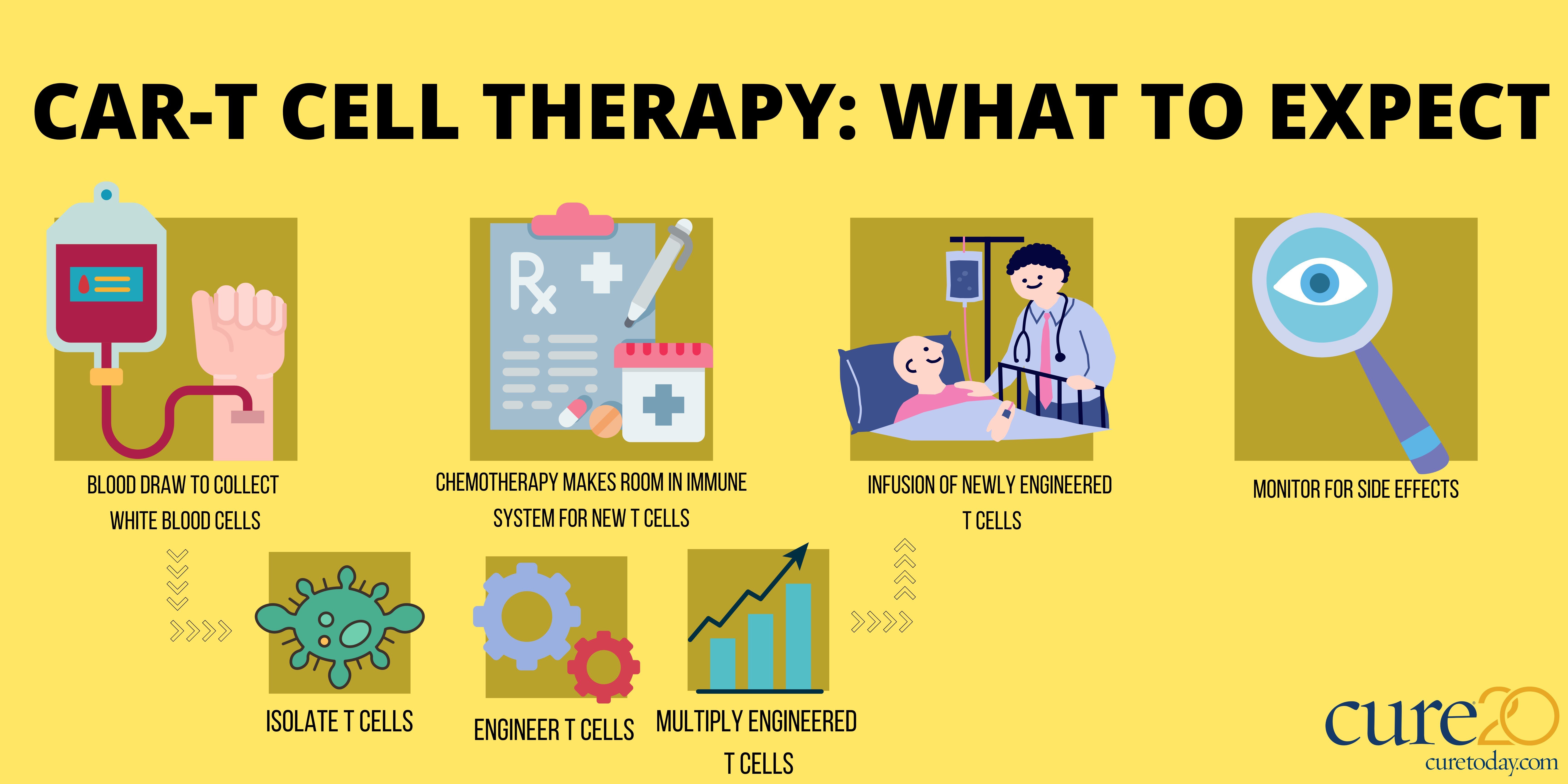
Breyanzi (lisocabtagene maraleucel), a CAR-T cell therapy, has shown promising results for patients with lymphoma in two studies, according to an announcement from pharmaceutical company, Bristol Myers Squibb.
Both studies — the TRANSCEND FL and TRANSCEND NHL 001 — met their main goal of overall response rate, which is the percentage of patients whose disease shrunk or disappeared as a result of treatment, Bristol Myers Squibb announced.
Breyanzi is a CAR-T cell thearpy that engineers patients' T cells to find and fight cancer.
“For people living with relapsed or refractory follicular lymphoma or mantle cell lymphoma, there are limited treatment options that provide deep and durable responses, especially for patients with high-risk disease,” Anne Kerber, senior vice president, head of cell therapy development, Bristol Myers Squibb, said in the news release. “These continued unmet needs coupled with our deep understanding of lymphoma biology drive us to deliver transformative treatments for patients. We believe these data further confirm Breyanzi’s best-in-class and best-in-disease profile and underscore the significant progress we are making in bringing the promise of our differentiated CAR-T cell therapy, Breyanzi, to more patients.”
The phase 2 TRANSCEND FL study evaluating Breyanzi in approximately 213 patients with follicular lymphoma started in July 2020, and is estimated to be completed by September 2028. The phase 1 Transcend NHL 001 study of 385 patients with non-Hodgkin lymphoma, which started in January 2016, had an estimated completion date of this month.
Bristol Myers Squibb reported that Breyanzi not only met the primary endpoint of overall response rate in both studies, as well as the secondary endpoint of complete response rate (when no signs of cancer are detected after treatment), and that no new safety signals for the treatment were reported in either study.
Bristol Myers Squibb plans to share detailed results from both studies “at an upcoming medical meeting” and discuss the findings with health authorities, according to the news release.
The United States Food and Drug Administration (FDA) previously approved Breyanzi for adults with large B-cell lymphoma (LBCL), including diffuse large B-cell lymphoma (DLBCL) not otherwise specified (including DLBCL arising from indolent lymphoma), high-grade B-cell lymphoma and primary mediastinal large B-cell lymphoma.
READ MORE: FDA Approves Breyanzi for Previously Treated Large B-Cell Lymphoma
Breyanzi has also been approved by the FDA for patients with follicular lymphoma grade 3B, who have refractory disease to first-line chemoimmunotherapy or relapse within 12 months of first-line chemoimmunotherapy, refractory disease to first-line chemoimmunotherapy or relapse after first-line chemoimmunotherapy and are not eligible for hematopoietic stem cell transplantation (HSCT) due to comorbidities or age or relapsed or refractory disease after two or more lines of systemic therapy, according to the FDA website.
Side effects of Breyanzi, according to Bristol Myers Squibb, have included cytokine release syndrome (an influx of inflammatory molecules, called cytokines) and neurological toxicities, resulting in the treatment being only available via a restricted program under BREYANZI REMS, a risk evaluation and mitigation strategy.
For more news on cancer updates, research and education, don’t forget to subscribe to CURE®’s newsletters here.