
It was disconcerting to me when my oncologist was unsure about the treatment he was giving me and knew what the next would be without giving the first a chance.

It was disconcerting to me when my oncologist was unsure about the treatment he was giving me and knew what the next would be without giving the first a chance.
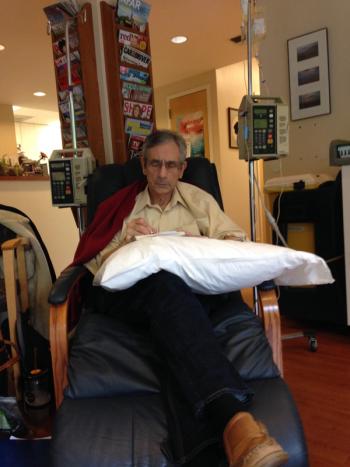
For the Jewish New Year, I had no more signs of lymphoma.
Even the difficult experiences that we share, like the emotional and physical discomfort that comes with a cancer scan, can be a little easier when we know someone else has been through them, too.
Treatment with Brukinsa and Gazyva poses a new option for certain patients with follicular lymphoma, with tolerable side effects, an expert told CURE®.

I've learned that there are many differing perspectives when it comes to treatment for cancer, especially from patients to researchers.

Last week we saw some FDA approvals come through, as well as research that explored the psychosocial outcomes of individuals who survived pediatric rhabdosarcoma.

The Food and Drug Administration approved Brukinsa plus Gazyva for patients with relapsed or refractory follicular lymphoma who have undergone two or more prior lines of therapy.

When I told my friends I started feeling better during cancer treatment, they were all so confused.

Having all of these screenings for cancer terrifies me, even though they could be almost certainly nothing.

Whenever I experience a new ache or patch of itchy skin, I immediately think it's a health issue — or a recurrence.
Researchers reported long-lasting responses and no new safety concerns in patients with relapsed/refractory follicular lymphoma during the 2023 ASH Annual Meeting and Exposition.

Please don’t tell me I have the “good” kind of cancer or comment on how much weight I’ve lost.

The Food and Drug Administration will speed up the review of odronextamab for patients with relasped or refractory follicular lymphoma or diffuse large B-cell lymphoma.

One patient was able to access a therapy for follicular lymphoma before it was approved by the FDA, which allowed him to help future patients through his experience.

Patients with relapsed/refractory follicular lymphoma — a group that traditionally has limited treatment options — now have a new bispecific antibody available.

The combination treatment demonstrated survival and safety results similar to those of chemotherapy for patients with advanced, untreated follicular lymphoma.

The FDA approval of Breyanzi, a novel CAR-T cell therapy, provides many patients with lymphoma a new treatment option that can be used earlier in the course of their disease, according to an expert.

The priority review comes following positive clinical trial results, which may lead to a shift in the treatment paradigm for follicular lymphoma, according to an expert.

Kymriah, a CAR-T cell therapy, was approved by the Food and Drug Administration for the treatment of patients with pretreated, relapsed/refractory follicular lymphoma.
Treatment with TRPH-222 led to lasting responses in certain patients with relapsed/refractory B-cell non-Hodgkin lymphomas, warranting a larger investigation into the drug, according to the researchers.
Kymriah may represent a more effective option than currently available treatments for patients with heavily pretreated follicular lymphoma, according to an expert from The University of Texas MD Anderson Cancer Center.
New findings show that 43% of patients with certain blood cancers produced COVID-19 antibodies after receiving a third full dose of the mRNA vaccine.

The Food and Drug Administration will review parsaclisib for the treatment of patients with follicular lymphoma, marginal zone lymphoma and mantle cell lymphoma.

Kymriah was granted a priority review for the treatment of pretreated patients with relapsed or refractory follicular lymphoma.

“We tried very hard, my daughter and I, to have a daughter-father relationship, not a daughter-doctor relationship,” said Dr. Michael Weiner on this episode of the “CURE® Talks Cancer” podcast, in which he discussed his career, his own cancer journey and his daughter’s diagnosis.