Kidney Cancer
Latest News
Video Series

Latest Videos
Shorts

Podcasts
More News
Receiving a diagnosis of stage 4 renal cell carcinoma can be overwhelming, but the treatment landscape has changed in recent years. Learn more from CURE.
As kidney cancer survival improves, more patients report cognitive changes affecting memory, fatigue, and overall mental clarity

Stage 3 kidney cancer can be managed with surgery, immunotherapy and targeted therapy; knowing your options helps guide informed decisions.

Although most patients with localized kidney cancer have high cancer-specific survival, surveys show a gap between doctor and patient perceptions of cure.
Dr. Michael Staehler highlighted nutrition’s role in kidney cancer, noting that diet, stress management, and lifestyle can impact outcomes and well-being.
The FDA has granted fast track designation to ITM-94 as a diagnostic agent for the detection of the most common type of kidney cancer.

A real-world study found TKIs were the most common therapy for adults with metastatic renal cell carcinoma after prior immune checkpoint inhibitor use.

A study at IKCS finds higher BMI after kidney cancer diagnosis improves survival, while significant weight loss predicts poorer outcomes.

American Indian and Alaskan Native individuals face some of the highest kidney cancer burdens in the United States.
Patients with advanced renal cell carcinoma whose disease has progressed on first-line therapies may benefit from the combination of Fruzaqla and Tyvyt.

Dr. Ravi Munver discusses how minimally invasive techniques allow patients to preserve kidney function and practical guidance for navigating treatment.

Remote monitoring has been found to be associated with improved postsurgical recovery for patients with cancer.

As September concludes, the landscape of GU oncology continues to evolve, with important developments in prostate, bladder, kidney and other cancers.
This guide on stage 2 kidney cancer covers key info, treatments and what to expect, helping you prepare for informed discussions with your care team.

Molecular testing helps distinguish kidney cancer subtypes, ensuring patients get accurate diagnoses and treatment plans, avoiding unnecessary surgery.

Dr. Ravi Munver sat down for an interview with CURE to discuss progress in kidney cancer care.

I reflect on how my family and friends showed love in different ways during the early days of my chronic cancer diagnosis.
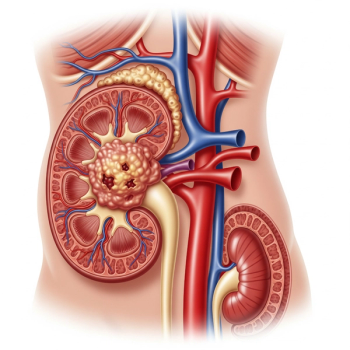
Researchers have created a novel immunotherapy for metastatic kidney cancer that could provide a new treatment option for patients with limited options.
Early-stage kidney cancer is limited to the kidney, usually under seven centimeters and often has a very good prognosis when treated promptly.

Kristen Dahlgren of the Cancer Vaccine Coalition spoke with CURE about why she remains hopeful for the future or cancer vaccines.

It is important to expand clinical trial access for patients with kidney cancer in order to ensure a representative study population.

Dr. Wenxin (Vincent) Xu discussed the benefit of using biomarkers to more precisely guide treatment for patients with renal cell carcinoma.

TAMs may help predict response to PD-1 inhibitors in metastatic clear cell kidney cancer, Dr. Berkay Simsek reported at the 2025 Kidney Cancer Summit.

Six-time survivor Staci Kirk founded the Stiletto Boss Foundation to provide community, support and practical help for women of color beyond treatment.

A dual cancer diagnosis rewired how I live, love, and show up in the world — with presence, purpose, and unapologetic passion.