News
Article
FDA Accepts Priority Review for Breyanzi in CLL
Author(s):
The Food and Drug Administration accepted a Priority Review for Breyanzi for the treatment of patients with relapsed or refractory chronic lymphocytic leukemia (CLL).
The Food and Drug Administration (FDA) recently accepted the supplemental Biologics License Application (sBLA) for Breyanzi (lisocabtagene mareleucel) to treat patients with relapsed or refractory chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who have received a Bruton tyrosine kinase inhibitor (BTKi) and B-cell lymphoma 2 inhibitor (BCL2i). Bristol Myers Squibb announced the FDA’s decision in a recent press release.
“Currently, there is no standard of care for people living with relapsed or refractory CLL or SLL after treatment with BTKi- and BCL2i-based regimens, leaving a critical unmet need for a treatment option that provides deep and lasting responses,” said Anne Kerber, senior vice president and head, Late Clinical Development, Hematology, Oncology, Cell Therapy (HOCT), at Bristol Myers Squibb. “This FDA acceptance brings us one step closer to offering these patients, for the first time, a personalized, T-cell based treatment option. We’re proud to further our commitment to bring the potential of CAR T cell therapy to more patients, building on Breyanzi’s foundation as a differentiated treatment option that has shown clinical benefit in the broadest array of B-cell malignancies.”
CAR-T cell therapies work by re-engineering patients' immune cells to find and fight cancer.
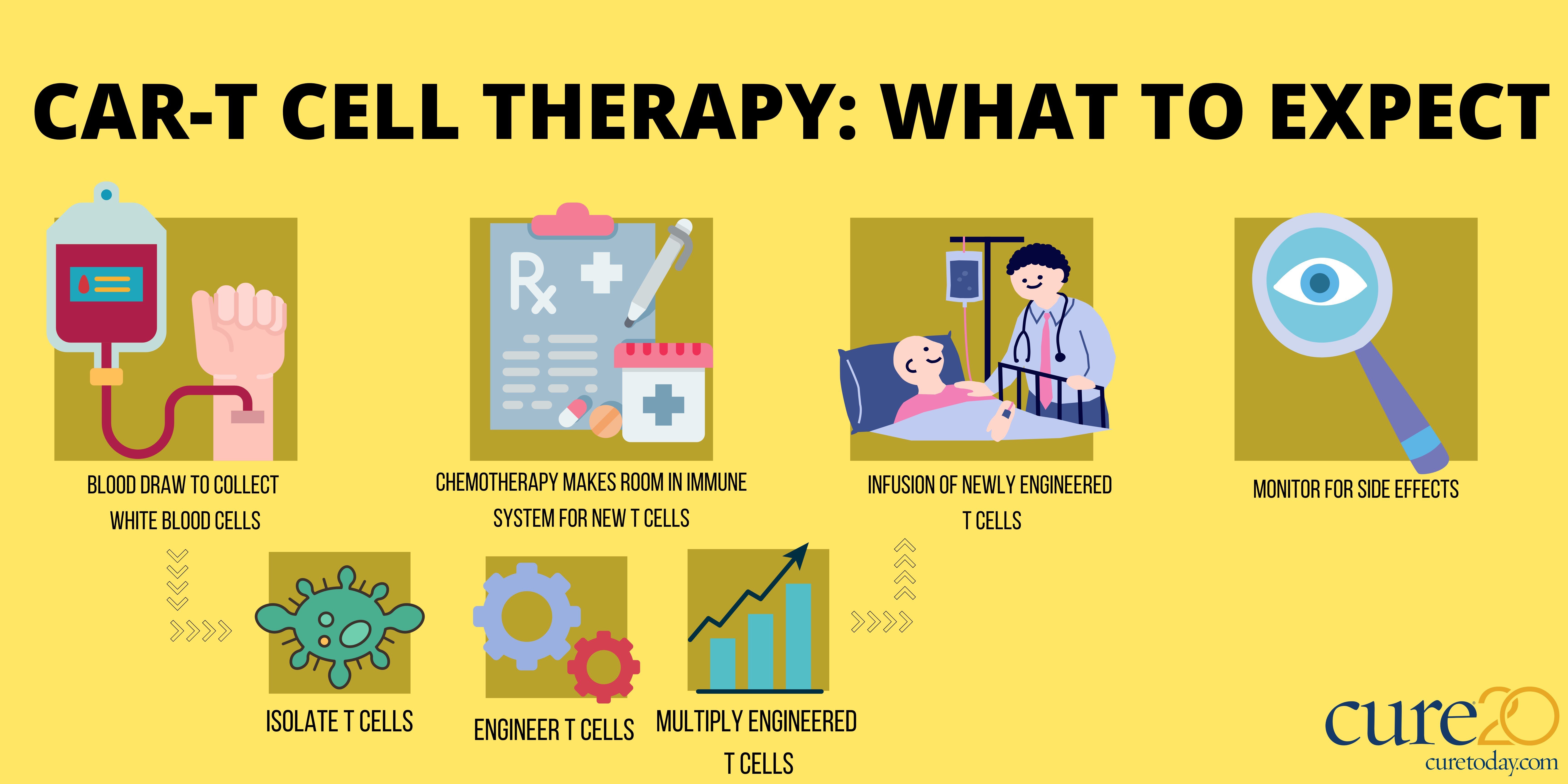
Breyanzi is created from a patient’s own T cells, which are genetically reengineering to find and fight cancer. The drug is currently FDA approved for refractory or relapsed large B-cell lymphoma after chemotherapy and for patients not eligible for stem cell transplant.
READ MORE:FDA’s Approval of Breyanzi Gives Some Patients With Lymphoma a ‘Life-Saving Therapy', Says Expert
The supplemental application was based on findings from the analysis of the TRANSCEND CLL 004 study, which was presented at the 2023 ASCO Annual Meeting. This became the first pivotal study that resulted in clinical benefit with a CD19-directed CAR-T cell therapy for patients with relapsed or refractory CLL, with BTKi and BCL2i treatment after progression.
TRANCSEND CLL 004 is a phase 1/2 trial that is analyzing Breyanzi in patients with CLL and SLL. Phase 1 on the trial focused on dose escalation to investigate its safety and efficiency, while phase 2 then focused on testing the ideal dose, which was found in the first phase of the trial. The primary endpoint for phase 2 is to conclude the complete response rate (percentage of patients whose disease disappears) and complete remission with bone marrow recovery.
If Breyanzi becomes approved by the goal date, this drug will become the first and only CSR T cell therapy for patients with relapsed or refractory chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who have received a BTKi and BCL2i.
The Prescription Drug User Fee Act (PDUFA) has a goal date of March 14,2024.
For more news on cancer updates, research and education, don’t forget to subscribe to CURE®’s newsletters here





