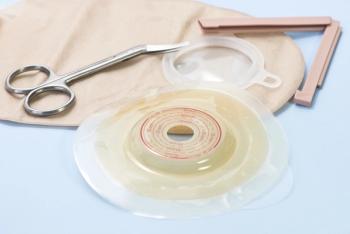
As part of CURE®’s Heal at Home series, we offer a guide on ostomies following surgery and how patients can best care for their stomas.

Ryan McDonald, Associate Editorial Director for CURE®, has been with the team since February 2020 and has previously covered medical news across several specialties prior to joining MJH Life Sciences. He is a graduate of Temple University, where he studied journalism and minored in political science and history. He considers himself a craft beer snob and would like to open a brewery in the future. During his spare time, he can be found rooting for all major Philadelphia sports teams. Follow Ryan on Twitter @RMcDonald11 or email him at [email protected].

As part of CURE®’s Heal at Home series, we offer a guide on ostomies following surgery and how patients can best care for their stomas.

As part of CURE®’s four-part gastrointestinal cancers webinar series, a panel of experts recently answered questions and offered advice around different types of testing.

CURE®’s Multiple Myeloma Heroes® Award Program honors and gives thanks to those who have made it their mission to change the lives of patients with multiple myeloma.

Watch Dr. Laura M. Spring, from Massachusetts General Hospital in Boston, discuss treatment in third line and beyond for hormone receptor-positive breast cancer, during the CURE® Educated Patient® Metastatic Breast Cancer Summit.

Watch Dr. Seth A. Wander, from Massachusetts General Hospital in Boston, discuss the first- and second-line treatment of hormone receptor-positive breast cancer, during the CURE® Educated Patient® Metastatic Breast Cancer Summit.

From a childhood cancer survivor throwing out the first pitch at Game 4 of the World Series to former WWE champion and actor Dwayne “The Rock” Johnson recording a message for a child with brain cancer, here’s what’s happening in the cancer space this week.

A woman without a family history of breast cancer shares how she went from volunteering at breast cancer walks to one day finding herself in the shoes of someone living with the disease.

Over the past 20 years, chemotherapy has remained the primary treatment for triple-negative breast cancer. However, recent advancements have shown that a one-size-fits all treatment approach no longer works.
The study investigators will review the efficacy of [Lu-177]-PNT2002, a radioligand treatment, compared with Zytiga or Xtandi in patients with metastatic castration-resistant prostate cancer.

The FDA granted a priority review to the oral drug quizartinib for the treatment of a form of acute myeloid leukemia, one of the more common types of leukemia.
Watch Dana Haagen, from Fox Chase Cancer Center, discuss clinical trials and how patients can join them, during the CURE® Educated Patient® Lung Cancer Summit.
Investigators of an expanded clinical trial have administered an investigational CAR-T cell therapy to the first patient with blood cancer.

Historically, there has been limited evidence to suggest the appropriate treatment sequencing of this melanoma patient population. However, new findings demonstrate starting with Opdivo plus Yervoy followed by Tafinlar in combination with Mekinist may be the best option.
The FDA granted a fast track designation to an investigational drug alone and in combination with Keytruda to treat patients with HPV recurrent/metastatic head and neck squamous cell carcinoma.

In 2018, 18 trekkers completed an awe-inspiring hike of the Himalayan highlands to the doorstep of the world’s tallest mountain, Mount Everest, to raise awareness and funding for multiple myeloma, a type of blood cancer. Three of the climbers discuss a bond that hasn’t broken.
The use of a multi-agent chemotherapy regimen resulted in improved outcomes in a group of children with pleuropulmonary blastoma, a rare form of lung cancer, when compared with other regimens.

Treatment with an investigational drug elicited a median progression-free survival (time from treatment until disease progression or death) of 8.9 months among patients with a form of unresectable or metastatic squamous non-small cell lung cancer, according to trial findings.

CURE®’s Lung Cancer Heroes® Award Program honors and gives thanks to those heroes who have positively affected the lives of patients with lung cancer.

Three out of four women with lung cancer reported experiencing moderate to severe sexual dysfunction, a survey shows.

Melanoma of the skin has been shown to be greatly associated with exposure to sunlight, whereas uveal melanoma — commonly known as cancer of the eye — is not.

Pets may be like family to some individuals but caring for them during illnesses such as cancer may force some to make a difficult decision.
None of the more than 100 evaluable patients with mismatch repair deficient colon cancer have experienced disease recurrence after treatment with presurgical Opdivo plus Yervoy, according to study results.
Delivering Sarclisa via an under-the-skin injection may improve the quality of life of patients with pretreated relapsed, refractory multiple myeloma, according to recent research.

An expert provides insight on how primary uveal melanoma is often treated and sheds some light on an unmet need in treating the eye cancer.

Patients with breast cancer who received post-mastectomy breast reconstruction had similar recurrence rates regardless of the timing of their reconstructive surgery, according to recent study findings.

Cervical cancer survivor Ginny Marable heard about a story of a fellow cervical cancer survivor, Tamika Felder, not being able to preserve her fertility. The story moved Marable so much that she and her husband donated their embryos to Felder.
A representative of the company developing the investigational CAR-T cell therapy noted that they hypothesize that the combination “has the potential to further enhance efficacy and provide patients with a chemotherapy-sparing, second-line treatment option.”
Investigators plan to analyze if treatment with an experimental, CAR-T cell therapy is safe and effective in a small group of patients with relapsed or refractory metastatic colorectal cancer.

The FDA has set a date to decide whether or not to approve momelotinib for myelofibrosis, a rare type of blood cancer.

A patient with metastatic breast cancer recalls the emotional experience of being among the thousands of attendees who gave a standing ovation in an auditorium in Chicago when the results of the phase 3 DESTINY-Breast04 trial — which recently led to the FDA approval of Enhertu in HER2-low disease — were presented at the 2022 ASCO Annual Meeting.

Published: December 12th 2022 | Updated:

Published: February 9th 2021 | Updated:
Published: June 6th 2021 | Updated:

Published: October 20th 2020 | Updated:

Published: March 24th 2021 | Updated:

Published: May 4th 2021 | Updated: