Kidney Cancer
Latest News

Zenocutuzumab ‘Offers Potential New Standard of Care’ for NRG1-Positive Solid Cancers
Latest Videos

More News

Laura Wood, M.S.N., RN, OCN, has "skill, dedication, boundless compassion and drive to improve the lives of those affected by kidney cancer."

Bryan Lewis, president of KidneyCAN, and Chung-Han Lee, MD, describe unmet needs and future perspectives in the treatment of renal cell carcinoma.
Meryl Uranga describes her path with metastatic clear cell renal cell carcinoma after first treatment, and how it has affected her outlook on her life and future.
A look at how to consider quality-of-life clinical trial and patient-reported data when treating clear cell renal cell carcinoma with combination therapies.

Christine Amoroso, B.S.N., RN, OCN, figured out the best way to deliver direct-to-kidney treatment for her patients.

Meryl Uranga describes her personal experience with managing the side effects of treatment for clear cell renal cell carcinoma.

An overview of side effects commonly seen with lenvatinib plus pembrolizumab for the treatment of clear cell renal cell carcinoma.
Watch Dr. Thomas Hutson, Dr. Saby George, Dr. Jeff Yorio and Louise Gunter answer questions about combination therapy, during the CURE Educated Patient Kidney Cancer Summit.

Watch Louise Gunter, from Texas Oncology, discuss IO/TKI-related side effects, during the CURE Educated Patient Kidney Cancer Summit.

Watch Dr. Jeff Yorio, from Texas Oncology, discuss treatment with the combination of immunotherapy and tyrosine kinase inhibitors, during the CURE Educated Patient Kidney Cancer Summit.

Watch Dr. Saby George, from Roswell Park Comprehensive Cancer Center, discuss treatment with the combination of immunotherapies, during the CURE Educated Patient Kidney Cancer Summit.
Watch Dr. Thomas Hutson, Louise Gunter and Meryl Uranga discuss communicating with a care team about treatment with immunotherapy combined with tyrosine kinase inhibitors, during the CURE Educated Patient Kidney Cancer Summit.

Watch Dr. Thomas Hutson, Louise Gunter and Meryl Uranga discuss side effects and dose reductions after treatment with immunotherapy combined with tyrosine kinase inhibitors, during the CURE Educated Patient Kidney Cancer Summit.

Watch Dr. Thomas Hutson, Louise Gunter and Meryl Uranga discuss the treatment of combining immunotherapy with tyrosine kinase inhibitors, during the CURE Educated Patient Kidney Cancer Summit.
Watch Dr. Thomas Hutson, Dr. Jose Karam, Dr, Naomi Haas, and Kiran Kehoe answer questions about frontline therapy during the CURE Educated Patient Kidney Cancer Summit.

Watch Dr. Naomi Haas, from Abramson Cancer Center, discuss adjuvant therapy options, during the CURE Educated Patient Kidney Cancer Summit.
An expert discusses the side effects patients with kidney cancer may experience from immunotherapy and tyrosine kinase inhibitor treatment, and how more research is needed in to ensure patients experience less side effects.
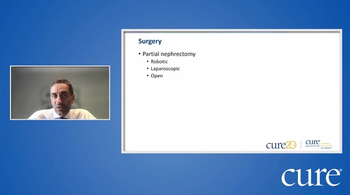
Watch Dr. Jose Karam, from The University of Texas MD Anderson Cancer Center, discuss surgical options, during the CURE Educated Patient Kidney Cancer Summit.

Clinical nurse Kiran Virdee explains why a multidisciplinary treatment approach to clear cell renal cell carcinoma is beneficial.

An oncologist describes the necessity of discussing options with a patient when picking a treatment approach for clear cell renal cell carcinoma.
Watch Dr. Vitaly Margulis, Dr. Rana Mckay and Kwame Garrett-Price answer questions about kidney cancer diagnoses during the CURE Educated Patient Kidney Cancer Summit.

Watch Dr. Rana R. McKay, from the University of California San Diego, discuss genetic testing in renal cell carcinoma during the CURE Educated Patient Kidney Cancer Summit.

The clinical development of BEMPEG plus Opdivo will end after clinical trials showed that the drug combination did not lead to significant improvements in outcomes in patients with kidney or bladder cancer.

A review of clinical trial data on combination treatments for clear cell renal cell carcinoma.

Patients with clear cell renal cell carcinoma speak to their experiences with systemic treatments.