Liver Cancer
Latest News
Video Series

Latest Videos
Shorts

Podcasts
More News

Dr. Anjana Pillai explains liver cancer types, risk factors, and prevention, stressing early screening for high-risk patients with liver disease.
This guide is designed to help you understand liver cancer and equip you with the right questions to ask your medical team.
The incidence of hepatocellular carcinoma has risen between 2010 and 2019; however, for people under 50 years of age, the proportion of cases has declined.

The FDA approved SIR-Spheres Y-90 resin microspheres for unresectable liver cancer, the only radioembolization approved in the U.S. for both HCC and mCRC.

Two people connect by sharing their experiences about taking care of their spouses who died from cancer, and the realization that sparked something more.

The FDA has approved Opdivo with Yervoy for the first-line treatment of adults with unresectable or metastatic hepatocellular carcinoma.
For patients with unresectable, non-metastatic hepatocellular carcinoma, Lenvima plus Keytruda in combination with TACE improved outcomes.

SBRT improved local progression-free survival over RFA in small recurrent HCC, particularly in tumors 2 cm or less.
Opdivo plus Yervoy shows efficacy and manageable safety as first-line treatment for unresectable hepatocellular carcinoma in the CheckMate 9DW study.

Stereotactic body radiation therapy combined with Nexavar showed a clinically important improvement in overall survival in locally advanced hepatocellular carcinoma.

Opdivo Qvantig received FDA approval for all previously approved indications in solid tumors.
Immune checkpoint inhibitor tumor regression may indicate which patients with hepatocellular carcinoma experience improved relapse-free survival.
For unresectable or metastatic liver cancer, the TPST-1120-301 trial of amezalpat combination therapy has received a “Study May Proceed” letter from the FDA.
From “Boy Meets World” actress Danielle Fishel discussing her breast cancer surgery to a young TikTok star dying from a rare liver cancer, here’s what’s happening in the cancer space this week.

Throughout September, the FDA approved drugs for the treatment of diseases including lung cancer, multiple myeloma, thyroid cancer and breast cancer.
In patients with unresectable liver cancer, treatment with Imjudo plus Imfinzi showed better survival outcomes compared with Nexavar.

Steatotic liver disease was associated with better liver function, lower recurrence rates, and improved overall survival in patients with liver cancer who received radiofrequency ablation.

Patients with intermediate-stage liver cancer may benefit from receiving a Lenvima-Keytruda combination with transarterial chemoembolization.

Tecentriq Hybreza, a subcutaneous delivery of the drug, was approved for certain types of sarcoma, in addition to lung, skin and liver cancers.

Through colon cancer and liver cancer, faith is what brought me to peace and clarity during treatment and now survivorship.
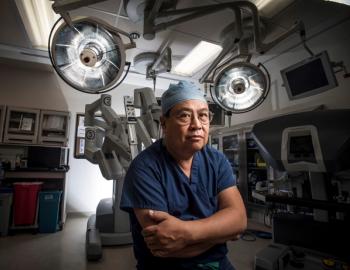
Patients can discuss minimally invasive robotic liver cancer surgery with their care teams. The process tends to be safe and effective, an expert said.

Even though I will never be cancer-free from cholangiocarcinoma, I’m ready to face the next chapter in my life and make the most of it.

Compared to open hemihepatectomy, the laparoscopic approach resulted in faster recovery time and higher quality of life, researchers found.

Jill May, B.S.N., RN, OCN, shows her commitment to her patients and their caregivers through acts of kindness and compassion — especially during tough times.
Adding Imfinzi and Avastin to transarterial chemoembolization led to a 23% reduction in the risk of progression or death in patients with liver cancer.