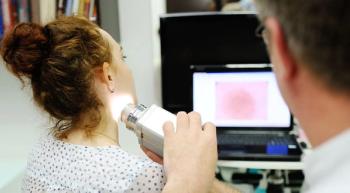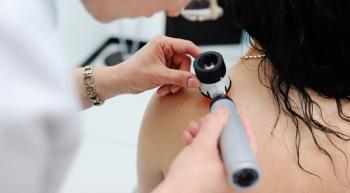
The panelists acknowledged that many individuals face barriers to receiving regular skin check-ups. Some people – especially those in rural areas – may not have a dermatologist near them. And even if they do make it in to a doctor’s office for a skin check-up, they may not be checked as thoroughly as they should be.